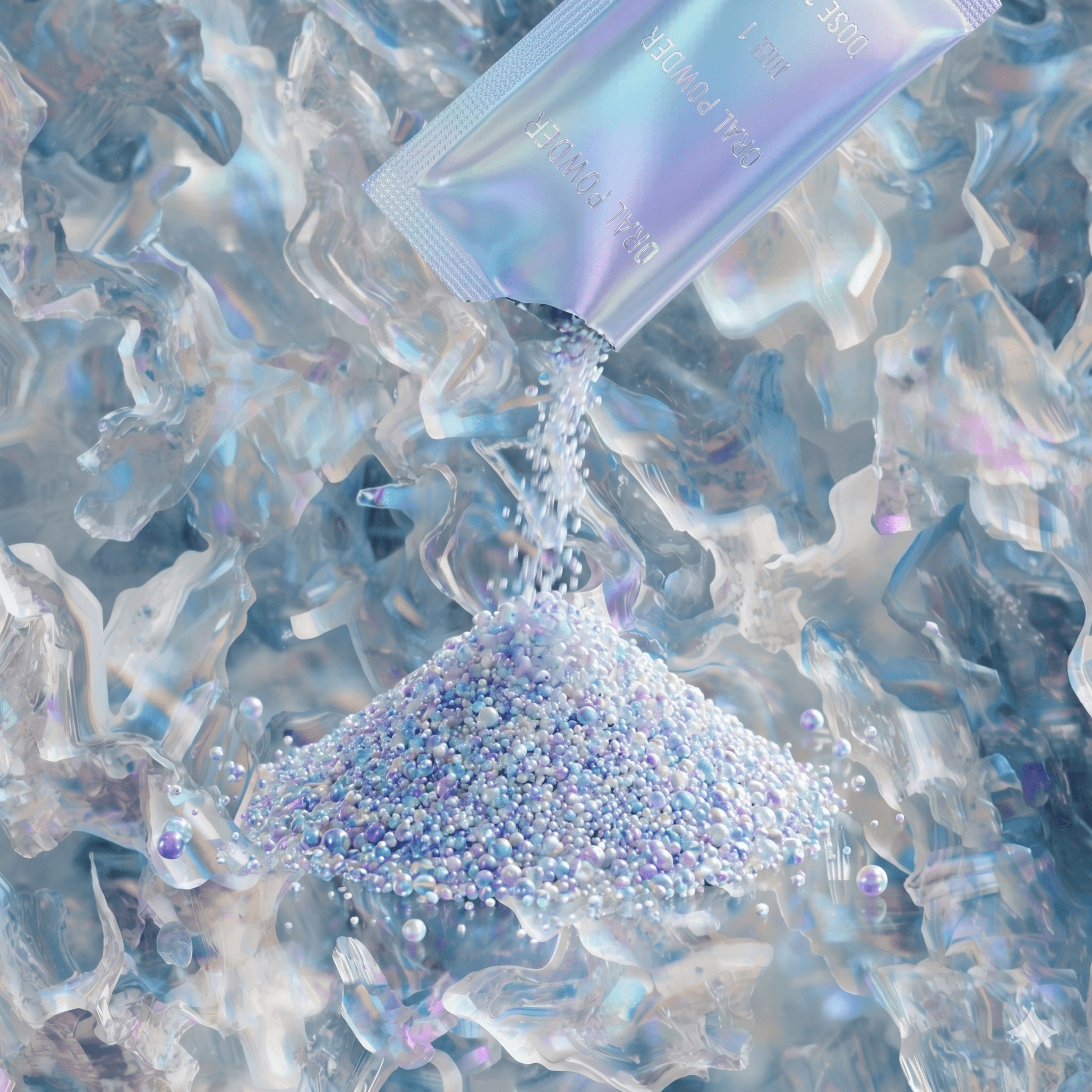Built on science, driven by the people behind the medicines we make.
Learn MoreFind products & categories quickly.
1 / 3
Which therapy area are you interested in?
Areas of Focus
Core pharmaceutical manufacturing, built for dependable production.
Oral solid (tablets, capsules, dry powder), oral liquid (syrups, suspensions), and external semi solid (ointments, creams, gels) dosage formulations, designed for consistency, quality, and scale under FDA Maharashtra licensing.
Manufacturing Scale
Manufacturing scale supported by structured systems.
Plant Premises
Built to support controlled pharmaceutical manufacturing.
The Meyonex plant premises are structured to support clean, controlled, and operationally disciplined pharmaceutical manufacturing. Site is engaged in manufacturing of Oral Solid Dosage Formulations which includes Tablets, Capsules & Dry Powder Preparations (General Category), Oral Liquid Formulations which includes Syrup & Suspension (General Category) and External Semi Solid Dosage Formulations which include Ointments, Creams & Gels (General Category) only which product licenses are issued by FDA, Maharashtra.
The facility includes fire hydrant systems, smoke detectors, fire extinguishers, emergency evacuation support, a dedicated effluent treatment system, epoxy-coated floors, and GMP doors fitted with view panels so activity can be monitored from clean room passages.
Premises planning
cGMP Norms
Effluent treatment support
Dedicated ETP
Environmental control
31 HVAC Units
Clean-room visibility
View-Panel Doors

Join the people behind disciplined manufacturing.
Help us deliver consistent, quality-first pharmaceutical manufacturing through strong documentation, operational rigor, and a culture that values ownership.
Leadership & Oversight
A cross-functional team built for disciplined manufacturing operations.
Photos use a compact crop; names and roles sit under each card. Where a full bio exists, hover the photo on desktop or open “View profile” on mobile.

Board of Directors
Mr. Arvind Kumar Sharma
Promoter & Group Chairman

Board of Directors
Mr. Arvind Awadhnath Sharma
Managing Director & Whole-Time Director
View profile
Mr. Arvind Awadhnath Sharma, our Promoter, Managing Director and Whole-Time Director holds Bachelor's degree in Engineering and Diploma in Business Management and has over 15 years of experience in the pharmaceuticals industry. He oversees manufacturing operations, procurement processes and marketing strategies of the company.

Board of Directors
Mr. Vinod Kumar Sharma
Whole Time Director & CFO
View profile
Mr. Vinod Kumar Sharma, our Promoter, Whole Time Director & CFO holds Master's degree in Business Administration (MBA) and has over 8 years of experience in the pharmaceuticals industry. He spearheads finance and accounts department of the company.
Board of Directors
Ms. Shivani Nagendra Sharma
Non-Executive Director
View profile
Ms. Shivani Nagendra Sharma, aged 25 years, is a Non-Executive Director of the Company. She completed her Bachelor of Arts in 2019 from Deen Dayal Upadhyaya Gorakhpur University, Gorakhpur. In the Company, she primarily serves in Admin and Human Resources, managing administrative functions and HR activities.